Description
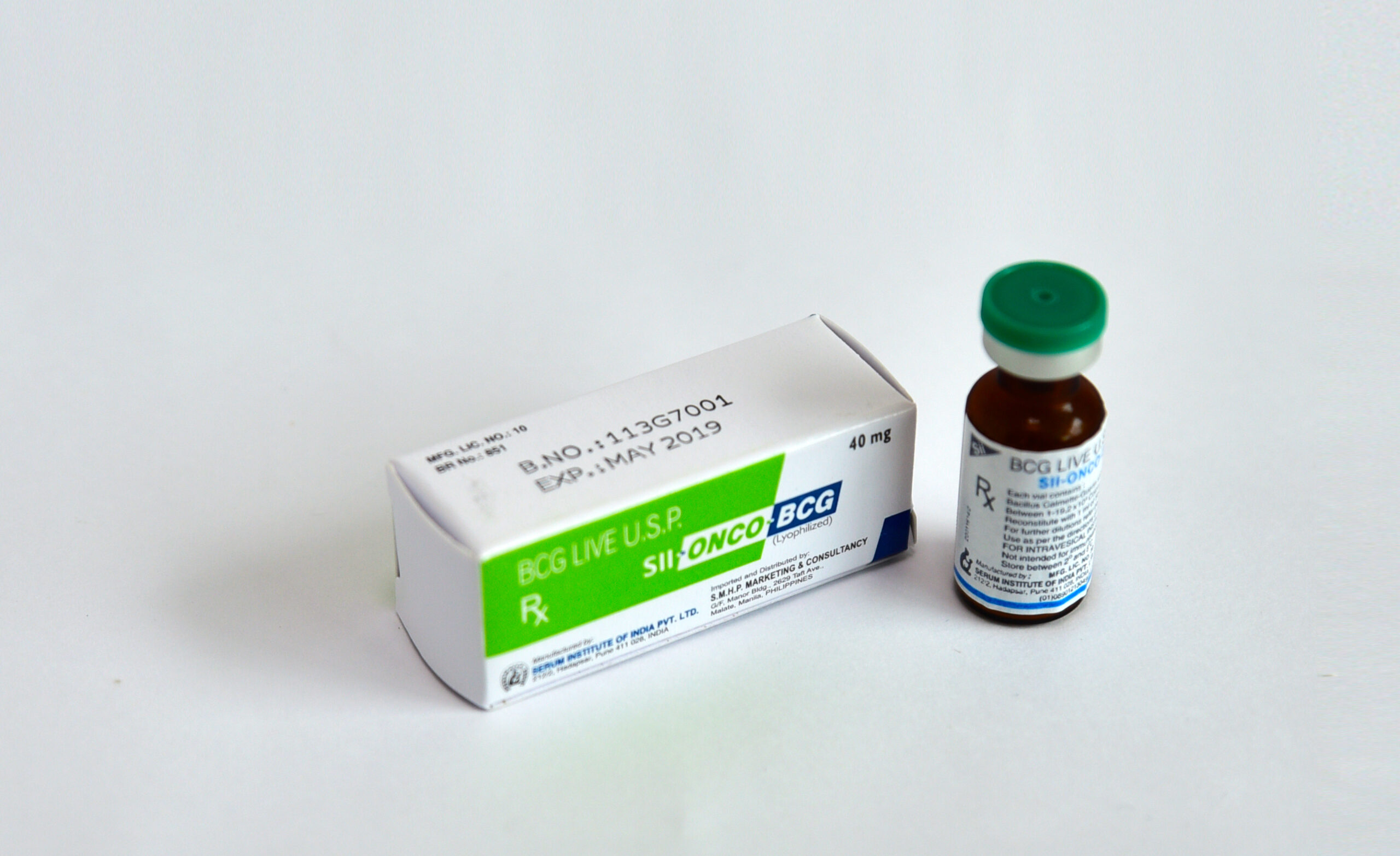
SII-ONCO-BCG (BCG LIVE USP) for intravesical instillation is a live lyophilized
preparation derived from attenuated strain of Mycobacterium bovis (Bacillus Calmette
Guerin).
COMPOSITION
Each vial contains:
Bacillus Calmette-Guerin strain: 40 mg/ml
Between 1-19.2 x 108 Colony Forming Units (CFU)
INDICATIONS
For treatment of flat Urothelial Cell Carcinoma in Situ of urinary bladder and as
adjunctive therapy following Transurethral resection of primary or relapsing superficial
noninvasive papillary tumors that are limited to the bladder mucosa (stage Ta or T1).
Intravesical BCG Immunotherapy has been shown to reduce tumor recurrence and
prevent progression.
DOSAGE
Treatment should be started 2-3 weeks after performing TURBT. The treatment
schedule is weekly repeated instillation with SII-ONCO-BCG (120 mg) during first 6
weeks, followed by 3 consecutive weekly instillations at 3 months, at 6 months and
thereafter every 6 months upto 36 months. This means that a patient who stays tumor
free after the initial resection will receive a total of 27 instillations in a period of three
years.
The duration and frequency of maintenance treatment should be evaluated on the
basis of tumor classification and clinical diagnosis.